Life
Happy Birthday To The First Oral Contraceptive!
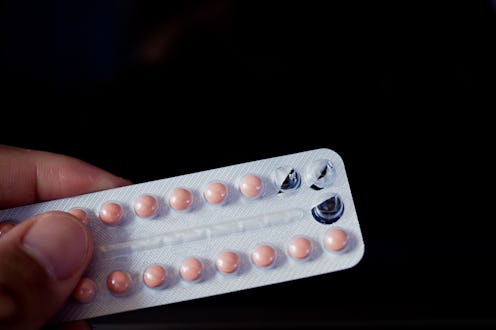
Fifty-six years ago, on May 9, 1960, the FDA approved the first oral contraceptive in the United States. The birth control pill would go on to change history. By allowing women to more fully control their fertility, the pill empowered them to have more control over other aspects of their lives, including sexual relationships, education, and employment. The history of birth control and the pill is a long one, characterized by fierce debates about reproductive freedom and the role of contraception in public life — debates that continue to play out to this day. To celebrate the anniversary of this history-making drug, let’s take a trip down memory lane:
This history of contraception stretches back many centuries. Some have suggested that in ancient Greece, the myth of Persephone eating only pomegranate seeds while in the underworld is a reference to the contraceptive properties of the pomegranate. For hundreds of years, people from all over the world have passed down recipes for herbal contraceptives, including a 13th-century man who would eventually become Pope after writing a book advising both men and women about the use of herbal contraception.
However, the quest for oral contraception as we now know it began in the mid-20th century. Birth control activist Margaret Sanger, who opened the first birth control clinic in the United States in 1916, followed and funded some of the scientific research on oral contraception in the 1940s and 50s. According to a report from Planned Parenthood, in the early 1950s, Sanger encouraged Katharine Dexter McCormick, an heiress and women’s rights activist, to fund further research by reproductive physiologists Gregory Pincus and Min Chueh Chang, who were making breakthroughs using synthetic progesterone as a contraceptive. McCormick would eventually donate $2 million to birth control research, the equivalent of more than $18 million when adjusted for today’s inflation.
Early clinical trials of the birth control pill were conducted by Dr. John Rock in Puerto Rico, starting in 1956. The researchers chose Puerto Rico as a testing ground for a number of reasons, including that it had a more female stable population than the United States, but a major one was that, in the mid-20th century, promoting contraception was illegal in most U.S. states, making clinical trials of contraception nearly impossible. A federal ban on contraception had been lifted in the 1930s, but laws against taking contraception or encouraging other people to use it still existed in most states.
In 1957, following successful clinical trials, the FDA approved the birth control pill as a treatment for “regulating menstruation.” Unsurprisingly, the number of women reporting irregular periods shot up, and by 1959, half a million American women were taking the pill for menstrual irregularity — and if they also appreciated the pill’s contraceptive side effects, well, who had to know?
Finally, in 1960, the FDA approved the sale of a drug called “Enovid” to be used as oral contraception. Despite being approved as contraception, promoting the pill as such was still illegal in many states. It wasn’t until 1965, when the Supreme Court ruled in Griswold vs. Connecticut that banning the use of contraception among married couples violated their constitutional right to privacy, that married women nationwide could openly seek oral contraception. (Single people had to wait until SCOTUS’s 1972 decision in Eisenstadt vs. Baird for the same right).
The launch of the birth control pill was not without controversy. One major issue was that early pills had very high levels of hormones, compared to similar medications today. Planned Parenthood reports that the first birth control pill, Enovid, contained 10,000 micrograms of progestin and 150 micrograms of estrogen; these days, a low-dose pill would contain between 50 and 150 micrograms of progestin and between 20-50 micrograms of estrogen. These high doses of hormones increased the likelihood that women would experience serious side effects of taking the pill, including heart attacks, stroke, and blood clots. Eventually these side effects led the FDA to require inserts in packages of birth control outlining possible risks, and to the development of lower dose pills. Despite these problems, the pill became an incredibly popular form of contraception: By 1967, 13 million women were on the pill worldwide. That number jumped to between 50 and 80 million in 1984 and more than 100 million today.
It's hard to measure to full impact that oral contraception has had on the world, if only because its influence is so far-reaching. The decades following the approval of the birth control pill have seen a dramatic reduction in maternal and infant deaths, as well as a decline in the rate of unwanted and mistimed births. Since 1965, the number of women participating in the U.S. labor force has nearly tripled, and between 1960 and 2013, the number of women completing four or more years of college rose from 5.8 to 37 percent. These changes in women’s education and their role in the workforce are due to a lot of different factors that have affected women’s rights and opportunities in the last 50 years, but women’s growing ability to make decisions about their reproductive lives was surely vital to that shift.
So let’s raise a glass, and toast the little pills that had — and continue to have — a big effect on history and women’s lives. Happy fifty-sixth birthday, Pill!
Images: Fotolia; Anastasia Zolotnitskaya / EyeEm/EyeEm/Getty Images; Giphy